July, 2014
- Carestream’s Digital DRX X-ray Detectors Surpass 9,000 Units
- 91 Hospital and Health System Layoffs in 2014
- HCA reports 14% rise in Q2 profit
Net income at Nashville, Tenn.-based Hospital Corporation of America rose 14 percent during the second quarter of this year, from $423 million in the second quarter of 2013 to $483 million this year.
- A CEO Checklist for High-Value Health Care
- Catholic health system ups its game
CHI expands its insurance business.
- Hospitals want fewer venders but Boston Scientific says they won’t be merging or buying
If scale is what is valued in the healthcare system these days, and hospitals want fewer and fewer vendors, then companies may be compelled to team up, whatever current strategy may be
- FDA Medical Device Guidances Issued in 2014’s Second Quarter
The Food and Drug Administration’s (FDA’s) Center for Devices and Radiological Health (CDRH) has issued a spat of guidance documents this quarter. here are some of the more significant FDA regulations that were issued in the second quarter of 2014. Provided below are brief summaries of several guidance documents that affect manufacturers and marketers of medical devices.
- Good times for Cerner’s salespeople
- Equipment and supplies sales to handle rising obesity rates increase
- GE Healthcare Sees Slight Profit Increase in Q2
The healthcare division reported revenue of $4.48 billion during the three months ended June 30, compared with $4.49 billion during the same three months in 2013. However, revenues increased from $726 million to $730 million.
- Are Hosptials using HIPAA to protect themselves instead of patients?
While HIPAA was enacted to protect the protected health information of patients, it appears hospitals are incorrectly citing HIPAA as a protection of their own interests, argues Charles Ornstein, a ProPublica healthcare journalist, in a report co-published on NPR.
- 10 CEOs: Why I chose healthcare
- 50 Top-Grossing Public Hospitals | 2014
- Portable DR a must to reduce turn around times
- Technology breakdowns a core strategic concern for hospitals
- Layoffs to continue – just be honest (not like Microsoft)
- Boston Scientific’s profits drop like a rock – 97%
- Diagnostic Imaging Utilization Trends and Reimbursement
At the peak in 2006, according to a 2008 Government Accounting Office report, Medicare spent $14.1 billion on diagnostic imaging. Since then, annual spending has steadily dropped, falling by 21 percent by 2010, based on a Neiman Policy Institute report. To date, this trend has not reversed, Levin said. - See more at: http://www.diagnosticimaging.com/reimbursement/imaging-utilization-trends-and-reimbursement?GUID=52331DD9-6AF6-4DA6-9850-104DD469DEE5&rememberme=1&ts=25072014#sthash.MPKxXUgQ.dpuf
- The current state of the Diagnostic Ultrasound market
- Are Lithium Batteries safe? UL establishes battery consensus standards for medical devices
UL (Underwriters Laboratories) has announced that the U.S. Food and Drug Administration (FDA) has recognized two UL battery safety standards as consensus standards for medical devices incorporating lithium or nickel-based batteries. The two standards are UL 2054 - Standard for Household and Commercial Batteries, and UL 1642 - Standard for Lithium Batteries (Cells).
- 15 Insights from Hospital & Health System CEOs on Leadership
- Medical Device User Fees Set to Drop in 2015, But Not by Much
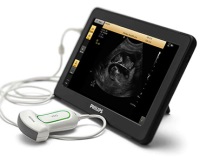
- Philips receives FDA 510(k) clearance for its ultra mobile VISIQ ultrasound system
- GE Healthcare faces economic, Obamacare challenges: Immelt
- 26 of the Most Controversial People in Healthcare
- The best Oncology programs in the US
If you sell into the Oncology space this is where you want to go.
- Universal Health Services – UHS – no profits but no debt either
- 10 Most-Used EHRs Among Physicians
- EHR – a new source of malpractice losses?
- Some Texas hospitals don’t have money as their owner goes on trial for fraud